Europe Biosimilars Market to Register 15.3% CAGR Between 2026 and 2033, Reports Persistence Market Research
Europe's biosimilars market is rapidly growing, driven by cost-effective alternatives, regulatory support, and expanding use across multiple therapeutic areas.
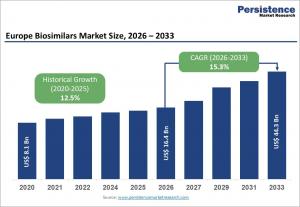
LONDON, UNITED KINGDOM, April 29, 2026 /EINPresswire.com/ -- The Europe biosimilars market is projected to witness remarkable growth over the coming years, with its market size estimated at US$ 16.4 billion in 2026 and expected to reach US$ 44.3 billion by 2033, growing at a CAGR of 15.3% between 2026 and 2033. Affordable alternatives to expensive biologic therapies are increasingly transforming the European healthcare landscape. The rising demand for cost-effective treatment options is driving the adoption of biosimilars across oncology, immunology, and rheumatology. Patent expirations of blockbuster biologics have opened opportunities for biosimilars to deliver substantial savings while maintaining comparable efficacy and safety profiles. Strong regulatory support from the European Medicines Agency (EMA) has accelerated approvals, facilitating wider adoption and integration into healthcare systems.
The adoption of biosimilars has generated cumulative savings exceeding €50 billion since 2006, including €10 billion in 2023 alone. Trends such as value-based healthcare, improved patient access, and expanded hospital pharmacy distribution are further propelling market growth. Continuous investment in research and development, along with increasing therapeutic options for chronic diseases, positions the Europe biosimilars market for sustained expansion through 2033.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/34923
Key Industry Highlights
The increasing patent expirations of biologics across therapeutic areas are expected to fuel biosimilars market growth. The preference for cost-effective biosimilar alternatives for treating autoimmune diseases, diabetes, and inflammatory conditions is likely to drive sales. Leading biosimilar manufacturers are enhancing digital strategies, including online marketing and distribution channels, to expand market reach. Oncology is projected to witness a CAGR of 13.7% by 2033, while hospital pharmacies, as a distribution channel, are expected to register a CAGR of 14.2%. Germany is likely to dominate the market with a CAGR of 15%, as major manufacturers such as Sandoz AG expand their portfolios across therapeutic areas.
Market Dynamics
Drivers – Wider Reimbursement and Pricing Approvals
Several European countries recognize the importance of biosimilars in controlling healthcare costs, leading to favorable reimbursement policies that ensure inclusion in public health programs. Pricing strategies making biosimilars affordable have increased market adoption and improved patient access. Wider reimbursement and pricing approvals have significantly contributed to Europe biosimilars market growth.
Government and Regulatory Support
The EMA provides clear regulatory pathways for biosimilars, ensuring rigorous safety, efficacy, and quality standards. As of 2024, 106 biosimilars have been approved in Europe, with 89 remaining commercially available. Many European governments promote biosimilar adoption across therapeutic areas, improving accessibility and building market confidence.
Restraints – Intellectual Property and Slow Adoption
Intellectual property and patent litigation present significant challenges. Complex patent portfolios of originator biologics can delay biosimilar entry due to legal disputes. Practices such as “evergreening” extend exclusivity, increasing legal costs and slowing market availability. Adoption remains slow in some countries due to conservative prescribing practices, lack of awareness, and minimal price differences between biologics and biosimilars. These factors limit growth and prevent patients from accessing cost-effective treatments.
Opportunities – Switching and Expansion
Biosimilar-reference biologic switching offers substantial opportunities for cost reduction, particularly in chronic disease management. Ten biologics are expected to lose IP protection over the next decade, creating potential €30 billion in opportunities between 2030 and 2032. Expansion into non-oncology therapeutic areas, such as autoimmune diseases, diabetes, and inflammatory disorders, also presents significant growth potential, supported by regulatory approval and physician confidence.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34923
Category-wise Analysis
Autoimmune diseases dominate the Europe biosimilars market, accounting for approximately 36% market share. High prevalence of rheumatoid arthritis, psoriasis, Crohn’s disease, and ankylosing spondylitis drives adoption, aided by reimbursement policies and physician familiarity. Oncology represents the fastest-growing segment, with a projected CAGR of 13.7%, driven by rising cancer incidence, patent expiries, and cost-saving opportunities.
Hospital pharmacies are the fastest-growing distribution channel, projected to register a CAGR of 14.2% between 2026 and 2033. Hospitals remain central to administering complex biologic treatments and enabling cost-effective substitution with biosimilars, supporting patient monitoring and adherence to regulatory guidelines.
Competitive Landscape
The Europe biosimilars market is competitive, dominated by established pharmaceutical giants and specialized biosimilar manufacturers, including Sandoz, Amgen, Samsung Biologics, Biogen, and Celltrion. These companies leverage R&D capabilities, regulatory expertise, and strategic collaborations to strengthen market presence. Competition is intensifying due to pricing pressures, patent disputes, and the need for product differentiation based on safety and efficacy.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34923
Market Segmentation
By Drug
Adalimumab
Bevacizumab
Trastuzumab
Rituximab
Infliximab
Etanercept
Epoetin alfa
Filgrastim
Other
By Drug Class
Antirheumatics
TNF Alfa Inhibitors
VEGF/VEGFR Inhibitors
HER2 Inhibitors
Selective Immunosuppressant’s
Interleukin Inhibitors
By Indication
Oncology Diseases
Autoimmune Diseases
Skin Disorders
Inflammatory Bowel Disease (IBD)
Rare Diseases
Others
By Distribution Channel
Hospital Pharmacies
Retail Pharmacies
Online Pharmacies
Key Developments
In November 2025, Sandoz launched Afqlir®, a biosimilar to Eylea® (aflibercept), in the UK, with expansion planned across Europe. In July 2024, Apotex Inc. partnered with Coherus Biosciences to license toripalimab for Canada, marking its first innovative oncology biologic. Several biosimilars, including Samsung Bioepis’s EKSUNBI, Formycon AG’s FYMSKINA, and Reddy Holding GmbH’s ITUXREDI, received EMA approval.
Companies Covered
Fresenius Kabi, Biogen Inc., Celltrion, Inc., Pfizer Inc., Amgen Inc., Samsung Bioepis Co., Ltd., Biocon, Viatris (Mylan Pharmaceuticals Inc.), Coherus BioSciences, Inc., Boehringer Ingelheim Pharmaceuticals, Inc., Eli Lilly and Company, Bio-Thera Solutions, Ltd.
The Europe biosimilars market is set for sustained growth driven by patent expiries, regulatory support, cost-effectiveness, and expansion into new therapeutic areas, positioning it as a transformative segment in European healthcare.
Read Related Reports:
Inhaled Anti-Infectives Market: The global inhaled anti-infectives market size is projected to be worth US$ 3.7 Bn in 2026 and grow to US$ 6.6 Bn by 2033, with an expected CAGR of 8.6%.
Orthopedic Braces and Supports Market: Global orthopedic braces and supports market to grow from US$4.2Bn in 2026 to US$6.7Bn by 2033, registering a 6.9% CAGR during 2026–2033 worldwide forecast.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.